DIAGNOSTIC WORKUP
Anti-Nephrin Antibodies: A New Biomarker For Autoimmune Podocytopathies
By Dr. Vinay Srinivasa
Nephrologist
South Coast Renal
Senior lecturer, Griffith University, Queensland, Australia
Introduction
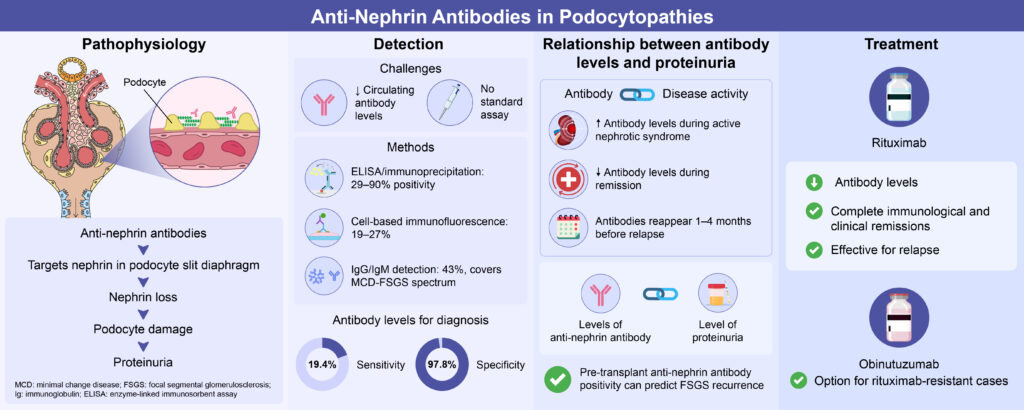
Minimal change disease (MCD), Focal segmental glomerulosclerosis (FSGS) and idiopathic nephrotic syndrome (INS) are important and common causes of nephrotic syndrome in adults and children respectively. Recently in a subset of this population group, autoantibodies targeting nephrin, a component of the slit diaphragm of podocytes, have been discovered. (9,20)
This important discovery has not only changed our conception of autoimmunity in podocytopathies but has also cultivated a view that these patients be reclassified under a novel subgroup; autoimmune podocytopathies. (7)
To diagnose MCD and FSGS in adults, a renal biopsy is required. In children with INS, renal biopsy is not always required. The characteristic histopathological findings on light microscopy in minimal change disease, show minimal or no significant changes to the glomeruli. In FSGS, sclerosis (scarring) affects a segment of the glomerulus and may not affect all glomeruli from the renal biopsy sample. In both conditions, diffuse podocyte effacement is seen on electron microscopy. The discovery of anti-nephrin antibodies may potentially serve as an non-invasive alternative to renal biopsy, although more research is needed.
Pathophysiology
Antibodies that target nephrin cause proteinuria. This phenomenon was first observed in animal models. (15,20, 21) Watts et al. were the first group to detect anti-nephrin antibodies in human subjects with minimal change disease. (23) In patients who underwent a renal biopsy, fine punctate immunoglobulin G staining on immunofluorescence and specific colocalization with nephrin was noted. (23)
Nephrin, a transmembrane cell adhesion molecule, is the core structural component of the slit diaphragm of podocytes. Its extracellular domains interact not only with itself but also with other proteins across the intercellular junction of podocyte foot processes. (1)
Nephrin functions by transducing signals to the podocyte interior and regulating cellular mechanisms that maintain its structural integrity. (8)
In disease states, nephrin transduces signals that cause self-deletion and removal from the intercellular junction by endocytosis. This disrupts the structural integrity of the slit diaphragm and results in proteinuria. (8,16)
Hengel et.al provided animal model evidence supporting a cause-and-effect relationship between anti-nephrin antibody levels and podocyte dysfunction, thereby proposing a potential pathogenic
mechanism. (10)
They modeled podocytopathy in mice by mimicking features consistent with human minimal change disease. They were able to achieve this by immunizing the mice with mouse nephrin to induce anti-nephrin autoantibody formation, which led to the development of nephrotic syndrome. Using proteomic and phosphoproteomic analyses of mice glomeruli immunized with nephrin, they critically showed that anti-nephrin antibodies exert direct effects on nephrin signaling and downstream pathways. Nephrin phosphorylation was also increased at the tyrosine residue Y1191, which is known to be involved in nephrin signaling and to play a key role in cytoskeletal and nephrin endocytosis.(10,21) Additional proteomic analysis of the immunized mice revealed increased levels of the adaptor proteins Shca (Shcl) and Clathrin (Cltc), which stimulate endocytic nephrin trafficking and downregulation of core slit diaphragm proteins.(10,12,22)
Further evidence to support the pathogenicity of anti-nephrin autoantibodies, was provided in a letter to the editor by Hengel and colleagues, published earlier this year. (9) Nephrin autoantibodies from a patient with minimal change disease were transfused to a rabbit. The rabbit developed proteinuria over a period of 5 days. Nephrin tyrosine phosphorylation was increased in response to human anti-nephrin autoantibodies in the injected rabbit. Precipitation of immunoglobulin G from the rabbit glomeruli followed by immunoprecipitation and western blotting revealed bound antibodies specific to rabbit nephrin, thereby supporting anti-nephrin antibodies as a causative factor. (9)
Evaluation of anti-nephrin antibodies
Although anti-nephrin antibody levels can be measured in serum, the detection methods remain controversial, posing questions about its pathogenicity. (5) Measurement of autoantibody titers using conventional immunoassays, such as western blotting, as in measuring PLA2R antibody levels in primary membranous nephropathy, are ineffective because of low circulating antibody levels. (10) Thus, a standardized assay is needed to measure antibody titers. Different detection methods have been employed with varying results.
Watts et al. evaluated patient sera for anti-nephrin antibodies using indirect enzyme-linked immunosorbent assay (ELISA) and immunoprecipitation of full-length nephrin from human glomerular extract or a recombinant purified extracellular domain of human nephrin. (23) Approximately 29% of patients with active minimal change disease, defined by a urine protein-to-creatinine ratio > 3 g/day, had detectable levels of circulating anti-nephrin antibodies. (21) Watts et al. believed that this low value was an underestimation. First, strict detection thresholds were enforced to prevent false-positive results; second, most of the study cohort had already received immunosuppressive therapy. (23)
By contrast, the patient cohort in the study by Hengel et al. had either untreated or relapsed disease, which was a significant difference from the population group in the study by Watts et al. (23) Hengel et al. measured anti-nephrin antibody levels using a sensitive hybrid immunoprecipitation ELISA method. (10) The anti-nephrin autoantibody status was determined by immunoprecipitation using the recombinant human nephrin ectodomain. The proteins obtained were incubated overnight with human serum or plasma before protein G was added. Immunoprecipitants were collected, electrophoresed, blotted, and analyzed for nephrin using an anti-nephrin antibody. As a result, the anti-nephrin antibody detected was exclusive to podocytopathies.
In adults with nephrotic syndrome due to minimal change disease who had not received immunosuppressive therapy, 69% tested positive for anti-nephrin antibodies. In untreated adolescents with idiopathic nephrotic syndrome, 90% were positive. Among those with relapsed disease previously treated with immunosuppression, 44% of adults with minimal change disease and 52% of adolescents with idiopathic nephrotic syndrome were positive. Only 9% of patients with primary FSGS showed anti-nephrin antibodies, while none were detected in patients with IgA nephropathy, lupus nephritis, or ANCA-associated vasculitis.
Chen et al. exclusively looking at a Chinese population, developed a new detection method using a cell-based indirect immunofluorescence assay to measure anti-nephrin antibody levels. Human nephrin was combined with a reference gene coding for green, fluorescent protein, which was inserted into a plasmid, and transfected into human embryonic kidney cells via lipofectamine 2000, a widely used transfection agent. This specific mode of gene delivery was designed to deliver nucleic acids into eukaryotic cells. (5) The cells were fixed onto a well plate, forming an antigen-substrate slide that could be used to detect circulating anti-nephrin antibody by indirect immunofluorescence method. (5)
In this study, the prevalence of circulating anti-nephrin antibody was shown to be 7/36 (19.44%) in patients with minimal change disease. Of the patients with minimal change disease with nephrotic range proteinuria, 7/26 patients (26.92%) tested positive for anti-nephrin antibodies, (5) which was like the results published by Watts et al. (22)
2/16 patients (12.5%) with FSGS tested positive for anti-nephrin antibody, suggestive of a potential spectrum disorder with MCD and idiopathic nephrotic syndrome. (5) Chen et al. used a receiver operating characteristic curve to assess the sensitivity and specificity of the antibody levels for diagnosis, obtaining a sensitivity of 19.4% and a high specificity of 97.8%. (5) Patients with Ig A nephropathy, diabetic nephropathy and primary membranous nephropathy were included in the study, these patients as expected, tested negative for anti-nephrin antibody. (5)
The above clinical studies primarily detected Immunoglobulin G (IgG) bound anti-nephrin antibody. A study by Shu et.al extended detection of anti-nephrin antibodies to include both IgG and IgM in large Chinese cohort of patients with both MCD and primary FSGS. (18) In this study, 256/596 patients were positive for anti-nephrin antibody (43%) (19) Of which, 30% (179/596) were positive for anti-nephrin IgG, 26.1% (155/596) were positive for IgM and 13.1% (78/596) were double positive for IgG and IgM (19)
Relationship between anti-nephrin antibody levels and proteinuria
Watts et al. and Hengel et al. established a correlation between antibody titers and proteinuria. Watts et al. first showed a reduction in circulating autoantibody levels during treatment response.(23) Serum samples were collected from 12 of 18 patients who tested positive for anti-nephrin autoantibodies when either complete remission (urine protein-to-creatinine ratio < 0.3 g/g) or partial remission was attained (>50% reduction in proteinuria).(23) The time to remission was equivalent in both the anti-nephrin antibody–positive and anti-nephrin antibody–negative groups. The relapse-free period after complete remission was shorter in the patients who were positive for anti-nephrin antibodies. (23)
Additionally, Hengel et al. demonstrated the longitudinal effects of anti-nephrin antibody titers during remission and relapse. The investigators examined 163 follow-up serum samples from 38 adult patients with minimal change disease or primary FSGS for circulating anti-nephrin autoantibodies by immunoprecipitation from samples without a prespecified follow-up time. Of the 38 patients,18 tested positive for anti-nephrin antibodies at different time points. Thirteen of the patients were adults with minimal change disease, and 5 had primary FSGS. As demonstrated by these findings, the presence or absence of anti-nephrin autoantibodies is associated with active disease and remission. Similarly, 33 samples from 22 children with idiopathic nephrotic syndrome were analyzed. Eighteen of the 22 children tested positive for anti-nephrin antibodies at different time points. Longitudinal paired samples were collected from 13 children with anti-nephrin antibody–associated idiopathic nephrotic syndrome at disease onset, before immunosuppressive treatment, and a few weeks after remission. All 13 patients were positive for anti-nephrin antibodies at onset, of whom 12 (92%) were negative for anti-nephrin antibody during remission. (10)
The study by Shu et.al, had 8 patients who had longitudinal follow-up. Serum samples were taken at time of clinical remission and at relapse. (19) 4 patients were diagnosed with diagnosed with IgG anti-nephrin antibody. 2 patients had MCD, and 2 patients had primary FSGS. (19)
The remaining 4 patients were diagnosed with IgM anti-nephrin antibody, 2 patients with MCD and 2 with primary FSGS (18) The levels of anti-nephrin antibody correlated with the level of proteinuria in all 8 patients. Interestingly, in 3 of the 8 patients, anti-nephrin antibodies reoccurred 1-4 months prior to relapse. (19)
Furthermore, a total of 39 patients positive for IgG anti-nephrin antibody (24 MCD and 15 primary FSGS) at diagnosis, had serum samples collected in remission. All the patients achieved clinical remission at the end of treatment and levels of IgG anti-nephrin were significantly lower at remission. (19)
22/24 patients with MCD (91.7%) and 13/15 patients with primary FSGS (86.7%) had immunological remission with negative anti-nephrin IgG at the time of clinical remission. (19)
In kidney transplant patients with FSGS, the detection and level of anti-nephrin antibodies before transplant can predict the risk of posttransplant recurrence. (18,23)
Precision therapy with Rituximab
Three case studies by Hengel et al. highlighted the relationship between autoantibody titers and treatment response to targeted B-cell depletion therapy. (10) In patients treated with B-cell depletion therapy, the treatment not only induced clinical remission but also reduced autoantibody levels. (7,10) The first case study involved a patient with relapsing minimal change disease. Anti-nephrin antibody titers fluctuated with proteinuria levels. The authors reported that partial treatment remission was achieved with prednisone and cyclosporine therapies. When rituximab was administered, complete and sustained remission occurred, as evidenced by the absence of both anti-nephrin antibodies in the serum samples and proteinuria (urine albumin-to-creatinine ratio < 0.3 g/g). (9) The other two patients were prospectively treated with rituximab therapy. One patient had minimal change disease, while the other patient had primary FSGS. Immunological and clinical remissions were successfully achieved.
Chen et al. reported similar observations in their study, where 4 of 7 patients with minimal change disease who were anti-nephrin autoantibody positive responded to rituximab therapy. However, the sample size was too small to show statistical significance. (5) The authors reported that patients positive for anti-nephrin autoantibodies had higher proteinuria at diagnosis, lower serum albumin levels, and higher triglyceride levels than those who were negative for anti-nephrin autoantibodies. This was suggestive of a more aggressive phenotype, indicating the need for a specific therapy. (5)
Similar findings were noted by Shu et.al (19) In this study, patients who tested positive for anti-nephrin antibody had nephrotic syndrome on presentation and were prone to frequent relapses. The authors concluded that anti-nephrin antibodies could serve as potential biomarkers for disease activity. (18)
Watts et al. also reported increased urine protein-to-creatinine ratios in patients positive for anti-nephrin autoantibodies compared with patients negative for anti-nephrin autoantibodies. However, these results were not statistically significant. (23)
Clinical implications
The detection of anti-nephrin autoantibodies may be considered as an alternative non-invasive diagnostic method in patients who are high-risk for renal biopsy complications. (7)
Moreover, patients who are anti-nephrin antibody positive have severe nephrotic syndrome, higher rates of relapse and shorter relapse free periods, indicating potential repeated dosage of targeted B cell depleting therapy. (19)
A recent case report by Loeffler et. al, has highlighted success in using Obinutuzumab in 2 patients with autoimmune podocytopathies who were resistant to Rituximab. Data using Obinutuzumab as a first-line treatment is scarce. More data is needed. (11)
The positive correlation between antibody levels and degree of proteinuria in all the clinical studies published suggest that it can be used a biomarker to monitor disease activity. (5,7,9,10,19)
Conclusion
Anti-nephrin antibodies have been shown to have high specificity in the diagnosis of patients with minimal change disease and correlate with disease activity. (5, 7,9,10,19) Not all patients with minimal change disease, primary FSGS, or idiopathic nephrotic syndrome will test positive for anti-nephrin antibodies. This suggests clinical heterogeneity. (5,7,10)
In patients who test positive for autoantibodies, precision treatment with rituximab therapy has shown favorable response. Thus, steroid toxicity is minimized. (7) Further studies are needed to examine the longitudinal course of anti-nephrin antibodies and treatment response to B-cell depletion therapy.
References
- Barletta, G.-M., Kovari, I. A., Verma, R. K., Kerjaschki, D., & Holzman, L. B. (2003). Nephrin and Neph1 co-localize at the podocyte foot process intercellular junction and form cis hetero-oligomers. Journal of Biological Chemistry, 278(21), 19266–19271. https://doi.org/10.1074/jbc.m301279200
- Basu, B. (2014). Ofatumumab for rituximab-resistant nephrotic syndrome. New England Journal of Medicine, 370(13), 1268–1270. https://doi.org/10.1056/nejmc1308488
- Basu, B., Sander, A., Roy, B., Preussler, S., Barua, S., Mahapatra, T. K. S., & Schaefer, F. (2018). Efficacy of rituximab vs tacrolimus in pediatric corticosteroid-dependent nephrotic syndrome. JAMA Pediatrics, 172(8), 757. https://doi.org/10.1001/jamapediatrics.2018.1323
- Cara-Fuentes, G., Clapp, W. L., Johnson, R. J., & Garin, E. H. (2016). Pathogenesis of proteinuria in idiopathic minimal change disease: Molecular mechanisms. Pediatric Nephrology, 31(12), 2179–2189. https://doi.org/10.1007/s00467-016-3379-4
- Chen, Q., Chen, S., Ye, Q., Lin, W., Liao, Y., Xiong, Y., Xu, J., Gao, R., Li, B., Liu, L., & Wei, L. (2025). Anti-nephrin antibody: A potential biomarker of minimal change disease. Clinical Kidney Journal, 18(3). https://doi.org/10.1093/ckj/sfaf012
- Fenoglio, R., Sciascia, S., Beltrame, G., Mesiano, P., Ferro, M., Quattrocchio, G., Menegatti, E., & Roccatello, D. (2018). Rituximab as a front-line therapy for adult-onset minimal change disease with nephrotic syndrome. Oncotarget, 9(48), 28799–28804. https://doi.org/10.18632/oncotarget.25612
- Fischman, C. J., & Holzman, L. B. (2025). Identification of nephrin autoantibodies signals new chapter for glomerular disease. American Journal of Kidney Diseases, 85(2), 262–265. https://doi.org/10.1053/j.ajkd.2024.09.001
- Garg, P., Verma, R., Nihalani, D., Johnstone, D. B., & Holzman, L. B. (2007). Neph1 cooperates with nephrin to transduce a signal that induces actin polymerization. Molecular and Cellular Biology, 27(24), 8698–8712. https://doi.org/10.1128/mcb.00948-07
- Hengel, F. E., Dehde, S., Kretz, O., Engesser, J., Zimmermann, T., Huber, T. B., & Tomas, N. M. (2025). Passive transfer of patient-derived anti-nephrin autoantibodies causes a podocytopathy with minimal change lesions. Journal of Clinical Investigation, 135(5). https://doi.org/10.1172/jci186769
- Hengel, F. E., Dehde, S., Lassé, M., Zahner, G., Seifert, L., Schnarre, A., Kretz, O., Demir, F., Pinnschmidt, H. O., Grahammer, F., Lucas, R., Mehner, L. M., Zimmermann, T., Billing, A. M., Oh, J., Mitrotti, A., Pontrelli, P., Debiec, H., Dossier, C., … Huber, T. B. (2024). Autoantibodies targeting nephrin in podocytopathies. New England Journal of Medicine, 391(5), 422–433. https://doi.org/10.1056/nejmoa2314471
- Loeffler T, Flahault A, Alla A, Frimat L, Kormann R. Obinutuzumab, an Anti-CD20, in Refractory Adult Autoimmune Podocytopathies: Report of 2 Cases. Kidney Med. 2025 Jun 24;7(9):101057. doi: 10.1016/j.xkme.2025.101057. PMID: 40837251; PMCID: PMC12361744
- Martin, C. E., New, L. A., Phippen, N. J., Keyvani Chahi, A., Mitro, A. E., Takano, T., Pawson, T., Blasutig, I. M., & Jones, N. (2020). Multivalent nephrin–Nck interactions define a threshold for clustering and tyrosine-dependent nephrin endocytosis. Journal of Cell Science, 133(4). https://doi.org/10.1242/jcs.236877
- Müller-Deile, J., Sarau, G., Kotb, A. M., Jaremenko, C., Rolle-Kampczyk, U. E., Daniel, C., Kalkhof, S., Christiansen, S. H., & Schiffer, M. (2021). Novel diagnostic and therapeutic techniques reveal changed metabolic profiles in recurrent focal segmental glomerulosclerosis. Scientific Reports, 11(1). https://doi.org/10.1038/s41598-021-83883-w
- Novelli, R., Benigni, A., & Remuzzi, G. (2018). The role of B7-1 in proteinuria of glomerular origin. Nature Reviews Nephrology, 14(9), 589–596. https://doi.org/10.1038/s41581-018-0037-z
- Orikasa, M., Matsui, K., Oite, T., & Shimizu, F. (1988). Massive proteinuria induced in rats by a single intravenous injection of a monoclonal antibody. The Journal of Immunology, 141(3), 807–814. https://doi.org/10.4049/jimmunol.141.3.807
- Qin, X.-S., Tsukaguchi, H., Shono, A., Yamamoto, A., Kurihara, H., & Doi, T. (2009). Phosphorylation of nephrin triggers its internalization by raft-mediated endocytosis. Journal of the American Society of Nephrology, 20(12), 2534–2545. https://doi.org/10.1681/asn.2009010011
- Ravani, P., Bonanni, A., Rossi, R., Caridi, G., & Ghiggeri, G. M. (2016). Anti-CD20 antibodies for idiopathic nephrotic syndrome in children. Clinical Journal of the American Society of Nephrology, 11(4), 710–720. https://doi.org/10.2215/cjn.08500815
- Shirai, Y., Miura, K., Ishizuka, K., Ando, T., Kanda, S., Hashimoto, J., Hamasaki, Y., Hotta, K., Ito, N., Honda, K., Tanabe, K., Takano, T., & Hattori, M. (2024). A multi-institutional study found a possible role of anti-nephrin antibodies in post-transplant focal segmental glomerulosclerosis recurrence. Kidney International, 105(3), 608–617. https://doi.org/10.1016/j.kint.2023.11.022
- Shu, Y., Huang, J., Jiang, L., Zhang, Y., Wang, F., Wang, X., Meng, L., Cheng, X., Liu, G., Wang, S., Zhao, M., Ronco, P., & Cui, Z. (2025). Anti-nephrin antibodies in adult Chinese patients with minimal change disease and primary focal segmental glomerulosclerosis. Kidney International. https://doi.org/10.1016/j.kint.2025.02.028
- Takeuchi, K., Naito, S., Kawashima, N., Ishigaki, N., Sano, T., Kamata, K., & Takeuchi, Y. (2017). New anti-nephrin antibody mediated podocyte injury model using a C57BL/6 mouse strain. Nephron, 138(1), 71–87. https://doi.org/10.1159/000479935
- Topham, P. S., Kawachi, H., Haydar, S. A., Chugh, S., Addona, T. A., Charron, K. B., Holzman, L. B., Shia, M., Shimizu, F., & Salant, D. J. (1999). Nephritogenic mAb 5-1-6 is directed at the extracellular domain of rat nephrin. Journal of Clinical Investigation, 104(11), 1559–1566. https://doi.org/10.1172/jci772
- Verma, R. (2006). Nephrin ectodomain engagement results in Src kinase activation, nephrin phosphorylation, Nck recruitment, and actin polymerization. Journal of Clinical Investigation, 116(5), 1346–1359. https://doi.org/10.1172/jci27414
- Watts, A. J. B., Keller, K. H., Lerner, G., Rosales, I., Collins, A. B., Sekulic, M., Waikar, S. S., Chandraker, A., Riella, L. V., Alexander, M. P., Troost, J. P., Chen, J., Fermin, D., Yee, J. L., Sampson, M. G., Beck, L. H., Henderson, J. M., Greka, A., Rennke, H. G., & Weins, A. (2022). Discovery of autoantibodies targeting nephrin in minimal change disease supports a novel autoimmune etiology. Journal of the American Society of Nephrology, 33(1), 238–252. https://doi.org/10.1681/asn.2021060794